In addition, there are a number of subclasses of IgG and IgA immunoglobulins for example, there are four human IgG subclasses (IgG1, IgG2, IgG3, and IgG4), having γ 1, γ 2, γ 3, andγ 4 heavy chains, respectively. IgA molecules have α chains, IgG molecules have γ chains, and so on. In mammals, there are five classes of antibodies, IgA, IgD, IgE, IgG, and IgM, each with its own class of heavy chain-α, δ, ε, γ, and μ, respectively. There Are Five Classes of Heavy Chains, Each With Different Biological Properties Although many die after several days, some survive in the bone marrow for months or years and continue to secrete antibodies into the blood. 
Plasma cells seem to have committed so much of their protein-synthesizing machinery to making antibody that they are incapable of further growth and division. Effector B cells can begin secreting antibody while they are still small lymphocytes, but the end stage of their maturation pathway is a large plasma cell (see Figure 24-7B), which continuously secretes antibodies at the astonishing rate of about 2000 molecules per second. 
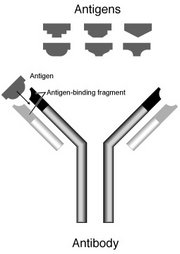
Such cells make and secrete large amounts of soluble (rather than membrane-bound) antibody, which has the same unique antigen-binding site as the cell-surface antibody that served earlier as the antigen receptor ( Figure 24-17). When a naïve or memory B cell is activated by antigen (with the aid of a helper T cell), it proliferates and differentiates into an antibody-secreting effector cell. As we discuss later, each of these receptors is stably associated with a complex of transmembrane proteins that activate intracellular signaling pathways when antigen binds to the receptor.Įach B cell produces a single species of antibody, each with a unique antigen- binding site. Each B cell has approximately 10 5 such receptors in its plasma membrane. Instead, they are inserted into the plasma membrane, where they serve as receptors for antigen. The first antibodies made by a newly formed B cell are not secreted. In this section, we discuss the structure and function of antibodies and how they interact with antigen.ī Cells Make Antibodies as Both Cell-Surface Receptors and Secreted MoleculesĪs predicted by the clonal selection theory, all antibody molecules made by an individual B cell have the same antigen- binding site. Mammals make five classes of antibodies, each of which mediates a characteristic biological response following antigen binding. Collectively called immunoglobulins (abbreviated as Ig), they are among the most abundant protein components in the blood, constituting about 20% of the total protein in plasma by weight. Synthesized exclusively by B cells, antibodies are produced in billions of forms, each with a different amino acid sequence and a different antigen- binding site. The white blood cells and activated complement components work together to attack the invaders. The binding of antibodies to invading pathogens also recruits various types of white blood cells and a system of blood proteins, collectively called complement (discussed in Chapter 25). Antibodies defend us against infection by binding to viruses and microbial toxins, thereby inactivating them (see Figure 24-2). Vertebrates inevitably die of infection if they are unable to make antibodies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
